A part leaves the washer bright and uniform, only to develop a faint orange haze minutes later. That rapid surface oxidation is known as flash rust. It most commonly appears after aqueous cleaning processes where water, heat, and freshly exposed metal meet in perfect chemical alignment. While it may look superficial, flash rust can disrupt coating adhesion, increase rework, and undermine quality control. Understanding why it forms and how to prevent it is essential for manufacturers using water-based parts washing systems.
What is flash rust?
Flash rust is rapid oxidation that forms on ferrous metals shortly after exposure to water and oxygen. Unlike long-term corrosion, which develops over extended environmental exposure, flash rust can appear within minutes or hours of cleaning.
The reaction is straightforward. When iron is exposed to oxygen in the presence of water, it forms iron oxide. In aqueous cleaning systems, parts are intentionally immersed or sprayed with heated water-based solutions. Once oils and soils are removed, the bare metal surface becomes highly reactive. If moisture remains and no corrosion protection is present, oxidation begins almost immediately.
The Science Behind Rust Formation
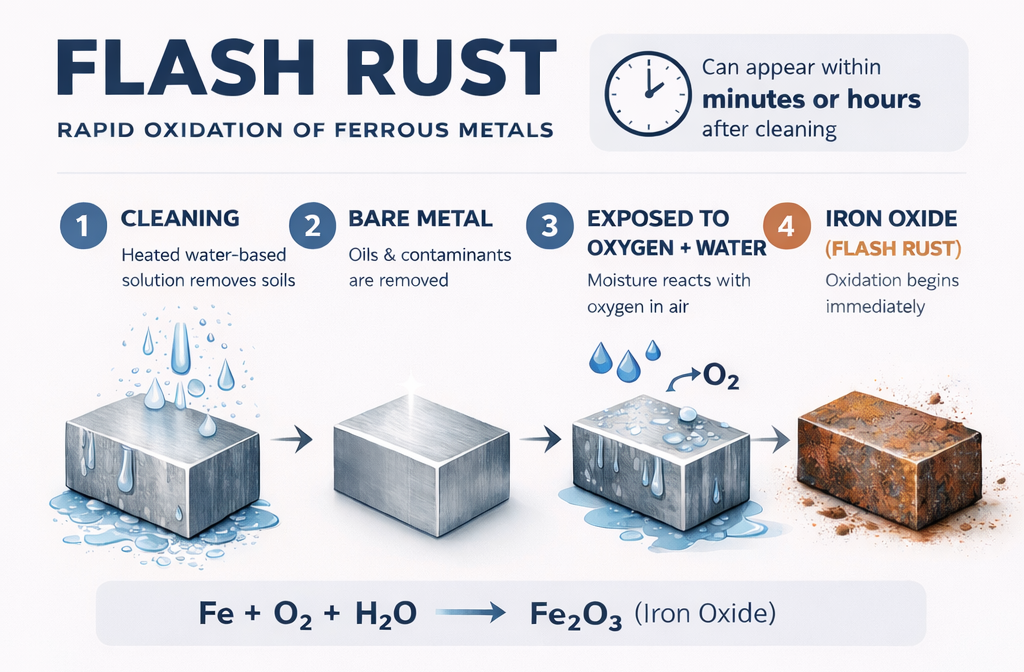
Water plays a central role in the corrosion process because it acts as an electrolyte. Even clean water contains dissolved oxygen and trace ions that enable electrochemical reactions. When moisture remains on a metal surface, it creates the conditions necessary for corrosion cells to form. Dissolved oxygen in rinse water accelerates this process, and residual moisture trapped in features such as blind holes, seams, or textured surfaces extends the time the reaction can occur.
Surface activation during cleaning further increases susceptibility. Aqueous detergents are designed to remove oils, coolants, and soils that may have previously acted as temporary corrosion barriers. Once these films are stripped away, the metal surface is left chemically active. Highly alkaline cleaners can enhance this reactivity. As a result, when the part exits the rinse stage, it is clean but vulnerable.
Temperature also influences flash rust formation. Elevated wash and rinse temperatures increase reaction rates. Warm parts retain heat as they leave the system, which can slow evaporation in humid environments and extend the time moisture remains on the surface. Higher temperatures also increase oxygen diffusion, supporting faster oxidation.
Water chemistry is another significant factor. Chlorides and dissolved salts increase conductivity, strengthening the electrolyte film left on the surface. Hard water minerals and improper pH balance can further accelerate corrosion. If corrosion inhibitors are absent or depleted, the system offers little resistance to oxidation once the part leaves the rinse stage.
Common Situations That Trigger Flash Rust
Flash rust often appears when process timing or chemistry control is inconsistent. Delays between rinsing and drying allow moisture to linger on exposed metal. Inadequate drying capacity can leave residual water in complex geometries. Poor rinse water quality, including elevated chlorides or high conductivity, increases corrosion potential. Facilities with high ambient humidity may unintentionally extend drying time. Carbon steel and cast iron are particularly susceptible, though stainless steel can also experience corrosion when chloride levels are elevated.
Why Flash Rust Is a Bigger Problem Than It Looks
Even light flash rust can create downstream challenges. Surface oxidation may interfere with coating or plating adhesion, leading to premature failures. Parts may require re-cleaning before finishing or assembly, increasing labor, water, and chemical consumption. Reject rates may rise if oxidation is misinterpreted as contamination or process instability. From a sustainability standpoint, rework cycles consume additional energy and resources, reducing overall manufacturing efficiency.
How To Prevent Flash Rust
Effective flash rust prevention begins with chemistry control. Many aqueous detergents include corrosion inhibitors that leave a temporary protective film on the metal surface. Maintaining proper concentration through regular monitoring ensures these inhibitors function as intended. If bath concentration drops or contaminants accumulate, corrosion protection may decline.
Water quality management is equally important. Evaluating rinse water for conductivity, hardness, and chloride content provides insight into corrosion risk. Using softened or deionized water in the final rinse stages reduces dissolved solids and lowers the electrolyte strength of any remaining moisture film. Cleaner rinse water decreases the likelihood of rapid oxidation.
Drying performance is another critical component. Heated air systems, high-velocity blow-off, or vacuum drying methods reduce residual moisture and shorten exposure time. Minimizing the interval between rinse and dry stages prevents extended contact between water and clean metal. The objective is to move the part quickly from a wet, reactive state to a dry, stable condition.
Consistent process control ties these elements together. Routine titration of cleaning solutions, monitoring inhibitor levels, preventive maintenance of nozzles and heaters, and evaluation of airflow systems all help ensure stable performance. In certain environments, controlling facility humidity may also be necessary to reduce corrosion risk.
Special considerations by material
Material type influences flash rust susceptibility. Carbon steel readily oxidizes when exposed to moisture and oxygen. Cast iron, with its higher surface area and porosity, can retain moisture and accelerate corrosion. Tool steels may require rapid drying and immediate protective measures. Stainless steel offers greater corrosion resistance but remains vulnerable in chloride-rich conditions. Mixed-metal assemblies require cleaning chemistries compatible with all substrates to avoid galvanic effects.
Designing a System That Prevents Flash Rust from the Start

The most reliable way to eliminate flash rust is to design the aqueous cleaning process with corrosion prevention in mind. Wash, rinse, and dry stages should function as an integrated system rather than isolated steps. Proper drainage, part orientation, balanced chemistry, controlled rinse water quality, and adequate drying capacity all contribute to consistent outcomes. Automation and standardized timing reduce variability that can lead to oxidation.
Clean Should Stay Clean
Flash rust is a predictable electrochemical reaction that occurs when freshly cleaned metal, moisture, oxygen, and insufficient protection intersect. It is not an unavoidable consequence of aqueous cleaning. By managing chemistry, controlling water quality, improving drying performance, and maintaining disciplined process controls, manufacturers can prevent flash rust and preserve both part quality and operational efficiency. Clean parts should leave the washer ready for the next operation, not beginning another cycle of rework.
Ready to learn more about our rotary basket part washers? Contact us to learn more or request free, no-obligation parts testing.

